Acetonitrile, also known as methyl cyanide, is a colorless liquid with a sweet, ether-like odor. It is a widely used solvent in various industries, including pharmaceuticals, agrochemicals, electronics, and analytical laboratories. Acetonitrile acn CAS 75-05-8 is known for its excellent solvency power, low viscosity, and high chemical stability.
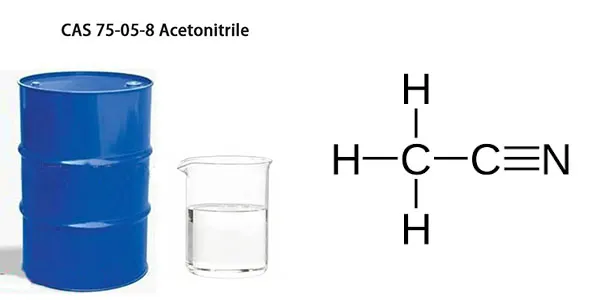
Acetonitrile Formula
Acetonitrile (CAS No. 75 05 8) is a hydrocyanic acid with the molecular formula CH₃CN.
Structure:
Acetonitrile CH3CN consists of two carbon atoms, three hydrogen atoms, and one nitrogen atom. The nitrile group (-CN) is attached to the methyl group (-CH₃), giving it unique reactivity and polarity.
Specification of Acetonitrile
| Property | Specification |
|---|---|
| CAS Number | 75-05-8 |
| Molecular Formula | CH3CN |
| Appearance | Colorless liquid |
| Odor | Sweet, ether-like |
| Purity | 99.9% min |
| Water Content | 0.05% max |
| Acidity (as acetic acid) | 0.01% max |
| Color (APHA) | 10 max |
| Boiling Point | 81.6°C |
| Melting Point | -45.7°C |
| Density (20°C) | 0.786 g/cm³ |
| Flash Point | 2°C |
| Solubility in Water | Miscible |
| Vapor Pressure | 97.6 mmHg at 20°C |
Want to know more about the properties and specifications of acetonitrile? Contact us!
Acetonitrile Uses
The uses of acetonitrile solvent are:
- Acetonitrile finds its application in copper refinings.
- It is used as a solvent for hydrocarbon extraction processes.
- It is used for manufacturing perfumes and other fragrances.
- It finds its application in the rubber industry.
- It is used in the pharmaceutical industry for making synthetic medicines.
- Acetonitrile (CAS 75 05 8) is used in electrochemical cells.
- Fatty acids are extracted from vegetable oils using acetonitrile.
Safety
GHS Hazard Statements:
- H225 (> 99.9%): Highly Flammable liquid and vapor [Danger Flammable liquids]
- H302 (99.8%): Harmful if swallowed [Warning Acute toxicity, oral]
- H312 (94.3%): Harmful in contact with skin [Warning Acute toxicity, dermal]
- H319 (99.8%): Causes serious eye irritation [Warning Serious eye damage/eye irritation]
- H332 (95.1%): Harmful if inhaled [Warning Acute toxicity, inhalation]
Handling and Storage
Storage conditions: Store it in an airtight container somewhere cool, dry and well ventilated, away from heat and other things it wouldn't mix with, like strong oxidisers.
Handling Precautions: Avoid inhalation and contact with skin and eyes. Use with adequate ventilation and proper protective equipment.
Frequently Asked Questions
Acetonitrile (CH3CN) is the simplest and one of the most stable nitriles.
Acetonitrile can neither be classified as an acid nor as a base.
Yes, Acetonitrile CH3CN is fully miscible with water at any ratio under normal ambient conditions.
Yes, acetonitrile solvent is toxic. It metabolizes in the body to produce hydrogen cyanide, which can cause irritation, confusion, and other health risks; severe exposure can even lead to death. Therefore, appropriate personal protective equipment must be used and operating procedures must be followed during handling.
Acetonitrile solvent should be stored:
- In a cool, well-ventilated area
- Away from oxidizing agents and ignition sources
- In tightly sealed containers
- According to hazardous chemical regulations (UN1648)
Contact Us:
Email: sales@gneebio.com
Website: https://www.gneebio.com/inquiry
Our company
TIANJIN GNEE BIOTECH CO., LTD. is a professional chemical raw materials supplier. We have passed quality system certification and have self-managed import and export rights. Our products are popular with customers. As a leading global supplier, we deliver certified chemical raw materials that comply with REACH, RoHS, ISO and other international standards. Our robust monthly production capacity ensures consistent quality and a reliable supply for bulk orders.


